Abstract
Irinotecan was approved by FDA as the first line therapy in combination with fluorouracil and leucovorin for metastatic colon or rectal cancer patients. Atropine is given for management of irinotecan-induced cholinergic syndrome, however, it is contraindicated for patient with angle-closure glaucoma. It is necessary to weigh the benefit before making a clinical decision. In this article, we are going to discuss whether it is appropriate to use atropine in colon cancer patients with history of angle-closure glaucoma before irinotecan therapy.
Keywords: colorectal cancer, irinotecan, atropine, angle-closure glaucoma
Introduction
Irinotecan was approved by FDA as the first line therapy in combination with fluorouracil and leucovorin for metastatic colon or rectal cancer patients. Irinotecan is also given as off-lable use for esophageal cancer, gastric cancer, pancreatic cancer, cervical cancer, ovarian cancer (recurrent), glioblastoma, Ewing sarcoma, rhabdomyosarcoma, non-small cell lung cancer, and small cell lung cancer1.
One of the most well-known side effects of irinotecan is cholinergic syndrome. Since a cholinergic syndrome rapidly occurs following high dose of irinotecan administration that can be ameliorate by atropine, this suggests that irinotecan directly interacts with acetylcholinesterase (AChE). And this hypothesis was confirmed by a series of detail kinetic analyses with irinotecan2. Diarrhea is one of the most concerned effect, the relationship between the pharmacokinetics of irinotecan and its metabolites (such as SN38) and diarrhea is also quite complex. Both irinotecan and its metabolite, SN-38 undergo enterohepatic circulation, and the concentration of SN-38 has been correlated with diarrhea in mice and in humans3. These side effects can be easily managed with atropine. Thus, atropine is widely used in irinotecan-containing chemotherapy regimens.
Incidence rate of cholinergic syndrome
Incidence rate of cholinergic syndrome, such as early diarrhea, perspiration, abdominal cramps, hiccup, visual accommodation disturbances, and lacrimation, ranges from 1.4% to 60% in patients receiving irinotecan4,5.
These side effects usually occur just after irinotecan infusion are not life-threatening and can be managed well in hospital, however, can affect the willingness of patients to receive the next course of chemotherapy.
Literature review: Atropine induced acute angle-closure glaucoma
Several case reports have mentioned about atropine induced acute angle-closure glaucoma in precipitated patients . They cause our concern of acute angle-closure glaucoma (AACG) when colon cancer patients are receiving atropine. A summary of these atropine-induced AACG case reports are listed on Table 1.
Table 1. Summary of atropine induced acute angle-closure glaucoma-case reports6,7
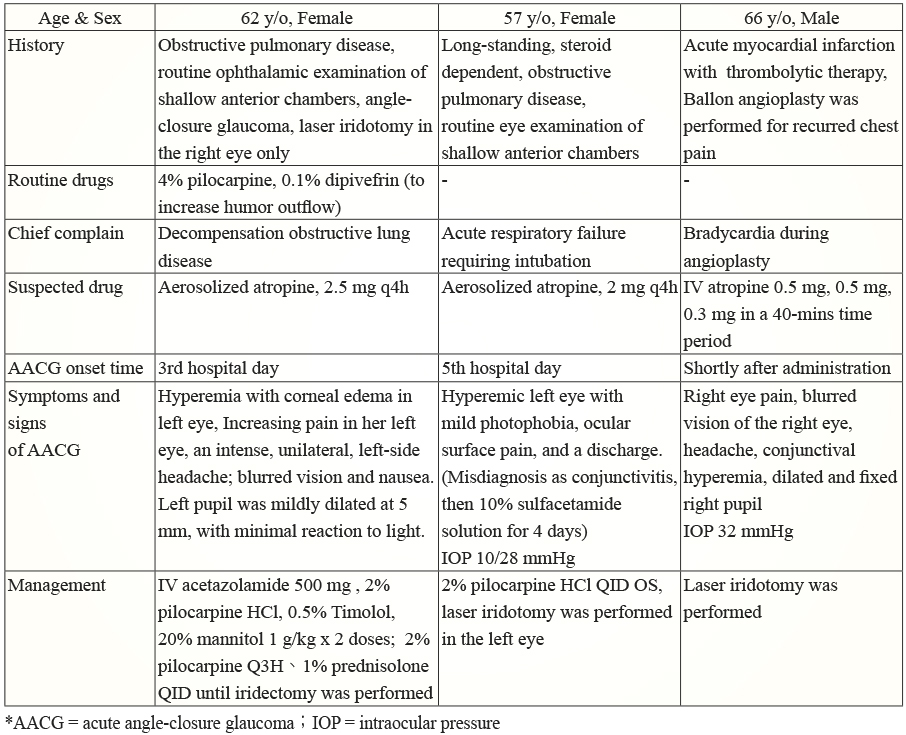
*AACG = acute angle-closure glaucoma;IOP = intraocular pressure
It is noteworthy that, an acute ACG attack was triggered in one patient by aerosolized atropine and oxybutynin caused another ACG attack, even though the patient received pilocarpine HCl miotic therapy routinely.
Dosing regimen of atropine for cholinergic syndrome
In Taiwan, atropine is used when patients suffer from cholinergic syndrome. In some hospitals, while it could be routinely prescribed at the first time and subsequent courses of chemotherapy. There are two administration routes to deliver atropine, intravenous and subcutaneous route. There are not any dosage difference in each route. The most consentaneous dosage of atropine is 0.5 mg and 0.25 mg. Though atropine dosage ranges from 0.2 mg to 2 mg regardless of administration route in literatures9,10. Should we use atropine for colon cancer patients receiving irinotecan with history of angle-closure glaucoma?
We proposed several reasons to support the use of atropine in colon cancer with history of angle closure glaucoma, and some reasons to hold atropine in these patients. These considerations are listed on Table 2.
Table 2. Factors should be considered in colon cancer patients going to use atropine with history of angle-closure glaucoma.

Alternative agents
Cheng et al. conducted a retrospective, single-center, nonrandomized, cohort study of adult patients (N = 80) treated with oral atropine 0.025 mg-diphenoxylate 0.25 mg or hyoscyamine as premedication before receiving irinotecan (total 532 cycles). Incidence of cholinergic syndrome did not differ between atropine-diphenoxylate and hyoscyamine groups after the first cycle or after the entire treatment course. There was a higher percentage of diarrhea but less abdominal pain or cramping and diaphoresis reported in the hyoscyamine group compared with the atropine-diphenoxylate group among patients who developed cholinergic syndrome. It is worth noting that additional premedications with intravenous atropine 0.5 mg were not required for subsequent irinotecan cycles in the hyoscyamine group, compared with the 23.3% of patients in the atropine-diphenoxylate group11. Theoretically, atropine-diphenoxylate and hyoscyamine both could result in enlarged pupils of the eyes and elevate ocular pressure. This article gave us a hint that two medications sharing similar mechanism could result in different side effect profile. Though the risk of atropine, diphenoxylate and hyoscyamine-induced AACG are still unknown. To date, there is not any research could answer which premedication is the best for patient with history of ACG before irinotecan infusion.
Conclusion
In summary, we didn't find any case report of subcutaneously administered atropine-induced AACG. However, it is unclear if low dose systemic atropine (< 0.6 mg) could cause ocular effects. Since incidence of cholinergic syndrome in not high, we recommend to hold atropine in colon cancer patient with history of angle-closure glaucoma who are going to for receive irinotecan, and to give a lower dose of atropine routinely if the patient suffers from cholinergic syndrome which may impede him to receive further courses of chemotherapy. We recommend that pilocarpine 2% eye drops could be given before starting atropine to reduced risk of AACG in colon cancer patients with history of angle-closure glaucoma as shown in Figure 14,6,7.
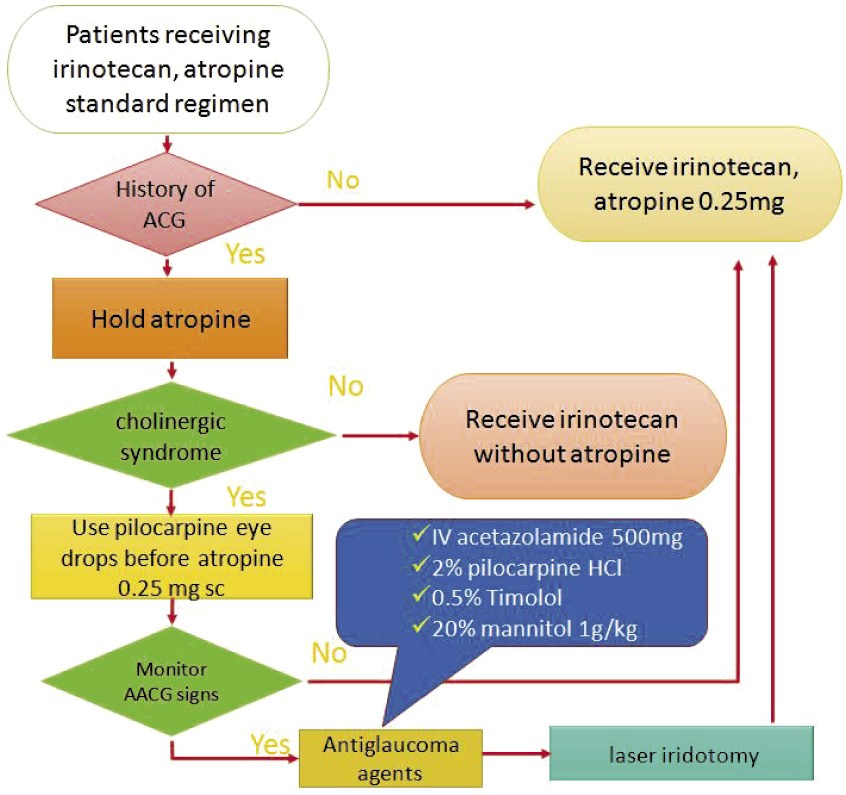
Figure 1. Flow chart of atropine use in colon cancer patients receiving irinotecan with history of angle-closure glaucoma
有閉鎖性青光眼病人接受 irinotecan 時使用 atropine 而誘發 AACG 的風險
成大醫院藥劑部藥師 陳弘益
摘要
美國 FDA 核准 irinotecan 與 fluorouracil 和 leucovorin 用於轉移性大腸直腸癌之第一線治療,atropine 則被用於緩解 irinotecan 引起的膽鹼症候群,然而 atropine 對於閉鎖性青光眼的病人而言屬於禁忌,使用前應評估好壞處的風險效益,本文我們將討論有閉鎖性青光眼病史的大腸直腸癌病人,預計接受 irinotecan 治療時,是否應該使用 atropine。
Reference:
1. Irinotecan: Drug information In: UpToDate, Post TW (Ed), UpToDate, Waltham, MA. (Accessed on January 19, 2015.)
2. Hyatt JL, Tsurkan L, Morton CL et al: Inhibition of acetylcholinesterase by the anticancer prodrug CPT-11. Chem Biol Interact. 2005;157-158:247-52.
3. Hecht JR. Gastrointestinal toxicity or irinotecan. Oncology (Williston Park). 1998;12(8 Suppl 6):72-8.
4. Package insert of Campto (irinotecan).
5. Abigerges D, Chabot GG, Armand JP, et al: Phase I and pharmacologic studies of the camptothecin analog irinotecan administered every 3 weeks in cancer patients. J Clin Oncol. 1995; 13(1):210-21.
6. Berdy GJ, Berdy SS, Odin LS, et al: Angle closure glaucoma precipitated by aerosolized atropine. Arch Intern Med 1991; 151:1658-1660.
7. Mandak JS, Minerva P, Wilson TW, et al: Angle closure glaucoma complicating systemic atropine use in the cardiac catheterization laboratory. Cathet Cardiovasc Diagn. 1996; 39:262-264.
8. Koch P, Grothaus-Pinke B, Hiddemann W, et al: Primary lymphoma of the stomach: three-year results of a prospective multicenter study. The German Multicenter Study Group on GI-NHL. Ann Oncol. 1997; 8 Suppl 1:85-8.
9. Gandia D, Abigerges D, Armand JP, et al: CPT-11-induced cholinergic effects in cancer patients. J Clin Oncol. 1993; 11(1):196-7.
10. Lai JS, Gangwani RA. Medication-induced acute angle closure attack. Hong Kong Med J 2012; 18:139-45.
11. Cheng C, Lau JE, Earl MA. Use of atropine-diphenoxylate compared with hyoscyamine to decrease rates of irinotecan-related cholinergic syndrome. J Community Support Oncol. 2015(1):3-7.

